Published: 13 March 2026. The English Chronicle Desk. The English Chronicle Online.
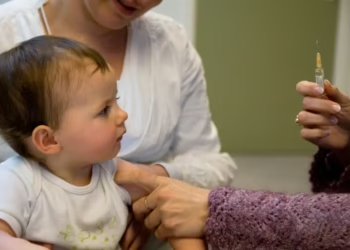
The phrase Canada plasma deaths has entered public debate after two donors died at private clinics. Authorities confirmed the incidents occurred while individuals donated plasma at centres operated by Grifols. Federal investigators have launched inquiries into the events, while critics question oversight of private plasma collection in Canada.
Health officials say fatal complications during plasma donation remain extremely rare across the country. However, the two deaths have reignited concerns about regulatory oversight and the growing role of private companies. Some critics argue the events reflect deeper challenges within Canada’s historically admired blood collection system.
According to Canada’s federal health regulator, Health Canada received reports describing “fatal adverse reactions” during plasma donations. The cases were reported following incidents in October 2025 and January 2026. Investigators confirmed both events involved donors experiencing severe distress during the donation procedure.
Both incidents occurred at clinics in Winnipeg, according to people familiar with the cases. Officials said they immediately visited the centres after learning about the reported fatalities. Early records suggested staff followed standard operating procedures during the donation processes.
The first donor was identified by friends as 22-year-old Rodiyat Alabede, an international student. She reportedly collapsed during a plasma donation in October 2025. Friends later organised an online fundraiser to support her family following the tragedy.
Those who knew Alabede described her as a compassionate student devoted to helping vulnerable communities. She had hoped to become a social worker after completing her studies. Friends said she believed strongly in service and helping others whenever possible.
The second fatal incident occurred roughly three months later at another clinic in the same city. Details about the individual have not been publicly disclosed. Authorities have said investigations remain ongoing as regulators review the circumstances surrounding both cases.
Representatives from Grifols said the company saw no immediate evidence connecting the deaths directly to plasma donation procedures. In a statement, the company stressed that donors undergo extensive health screening before participating in plasma collection.
Plasma, the pale yellow liquid portion of blood, plays a crucial role in modern medicine. It contains proteins essential for creating treatments used worldwide. Doctors rely on plasma-derived medicines to treat conditions including haemophilia, immune disorders, and severe burns.
Demand for these treatments has grown significantly in recent years, increasing pressure on plasma collection systems globally. Countries without sufficient domestic supply must often import plasma-derived products from abroad. Canada is among those countries that depend heavily on foreign plasma sources.
Officials say the nation does not collect enough plasma domestically to meet growing medical demand. Much of the supply comes from the United States, where paid donation systems are common. This dependence has led policymakers to consider expanding domestic plasma collection capacity.
Private companies have increasingly entered the sector as Canada tries to strengthen its supply chain. Critics argue that allowing commercial firms to operate plasma clinics creates potential conflicts between public safety and profit incentives.
Federal inspection reports show regulators have previously raised concerns about several facilities operated by Grifols. In one inspection conducted earlier this year, officials examined a centre in Saskatchewan.
Inspectors concluded that the site had failed to properly assess donor suitability during screenings. The inspection also identified eleven separate deficiencies related to operational procedures. As a result, regulators classified the facility as non-compliant under Canada’s blood regulations.
The findings highlighted weaknesses in equipment maintenance and documentation practices. Some records were incomplete or difficult to retrieve during the review. Inspectors also questioned the calibration and cleaning schedules of several machines used in the plasma collection process.
Another inspection conducted in Alberta identified ten similar deficiencies. These included issues related to donor screening, record keeping, and equipment oversight. The concerns raised further questions about consistent safety standards across private facilities.
Company representatives acknowledged the findings but emphasised that non-compliance ratings often involve procedural improvements rather than immediate safety threats. They said corrective action plans were submitted promptly to regulators. Implementation of those measures, they added, had already begun.
Officials confirmed clinics in Calgary and Regina remain open during the corrective process. Authorities continue monitoring the facilities closely as improvements are implemented.
Despite those assurances, the Canada plasma deaths have intensified scrutiny from advocacy groups and patient safety organisations. Critics argue the incidents highlight potential vulnerabilities within the country’s oversight framework.
Curtis Brandell, a blood safety advocate associated with the Canadian Hemophilia Society, said the deaths were deeply alarming. He noted that plasma donation has long been considered a safe medical procedure. Hearing about two deaths in the same city, he explained, raised immediate concerns.
Brandell said the events triggered memories of Canada’s historic blood contamination crisis decades earlier. During the 1980s and early 1990s, thousands of Canadians were infected with HIV or hepatitis C through contaminated blood products.
The scandal became one of the most significant public health failures in Canadian history. It ultimately led to a major national investigation known as the Royal Commission of Inquiry on the Blood System in Canada.
That inquiry recommended sweeping reforms aimed at improving transparency, safety standards, and regulatory accountability. Investigators also concluded that voluntary blood donation systems were safer and more ethical than commercial models.
Canada later introduced extensive reforms based on those recommendations. The modern blood system now operates under strict regulatory oversight designed to prevent similar tragedies.
Today the national blood supply is managed primarily by Canadian Blood Services. The organisation oversees donation programs, manages supply chains, and ensures safety standards are maintained nationwide.
However, the organisation does not currently collect enough plasma domestically for medical use. As demand for immunoglobulin treatments rises globally, policymakers have struggled to balance supply needs with ethical concerns surrounding paid donation.
Some provinces maintain strict rules against paying donors. Others permit private operators to collect plasma under regulated agreements. These differing policies have created a patchwork system across the country.
In provinces including Ontario, British Columbia, and Quebec, paid plasma donations are officially restricted. Yet exceptions exist where private operators work alongside public authorities.
Under agreements introduced in recent years, private clinics sometimes operate as agents for the national blood system. These arrangements allow plasma collection while technically maintaining compliance with provincial regulations.
Critics argue such partnerships blur the line between public and private healthcare services. They worry that oversight could weaken if commercial interests gain stronger influence within the blood supply network.
The Canada plasma deaths have therefore sparked broader questions about transparency and accountability. Advocacy groups say public trust depends on clear communication from regulators and healthcare organisations.
Investigators are now examining whether equipment, procedures, or screening methods contributed to the tragedies. Authorities have not yet confirmed any direct cause linking plasma donation to the fatalities.
Meanwhile, experts emphasise that plasma donation remains widely considered safe for healthy individuals. Millions of donations occur each year worldwide with minimal complications.
Even so, the recent Canada plasma deaths serve as a reminder that strong oversight remains essential. Healthcare systems must continuously review procedures to maintain safety and public confidence.
As investigations continue, families, regulators, and advocacy groups are seeking answers. The findings may ultimately shape future policies governing plasma collection across Canada.
For many observers, the tragedy highlights a deeper challenge facing modern healthcare systems. Balancing commercial efficiency with strict public accountability remains an ongoing task for policymakers everywhere.
The final conclusions of federal investigators may determine whether regulatory reforms become necessary. Until then, the debate surrounding the Canada plasma deaths continues across the country.