Published: 22 April 2026. The English Chronicle Desk. The English Chronicle Online
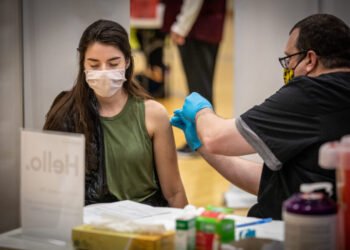
In a pivotal moment for global public health, researchers in the United Kingdom and the United States have officially commenced the first large-scale human trials for an investigational mRNA vaccine designed to combat the H5N1 avian influenza virus. The study, which delivered its first doses on Wednesday, represents a proactive “insurance policy” against a pathogen that experts warn possesses significant pandemic potential. By utilizing the same messenger RNA technology that revolutionized the response to COVID-19, scientists hope to provide a rapid and scalable defense mechanism should the virus, which currently ravages bird populations and dairy herds, adapt for sustained human-to-human transmission.
The trial centers on a vaccine candidate known as mRNA-1018, developed by the biotechnology firm Moderna in collaboration with the UK Health Security Agency (UKHSA) and the National Institute for Health and Care Research (NIHR). Unlike traditional influenza vaccines, which often rely on slow, egg-based manufacturing processes, the mRNA platform allows for the rapid synthesis of viral proteins within the human body to trigger a targeted immune response. This Phase 3 study aims to evaluate the safety and immunogenicity of the shot in approximately 4,000 healthy adult volunteers. In the UK alone, 3,000 participants—including many over the age of 65—will receive two doses of the vaccine across 26 clinical sites spanning England and Scotland.
The urgency of the trial is underscored by the changing nature of the H5N1 strain. While human infections remain rare and are almost exclusively linked to direct contact with infected animals, the virus has shown an alarming ability to jump between diverse mammalian species, including minks, marine mammals, and, most recently, dairy cows in the United States. Since 2024, more than 116 confirmed human cases have been recorded globally. Public health officials, including UKHSA Director Dr. Richard Pebody, emphasized that while the current risk to the general population remains low, the “real possibility” of the virus evolving to move easily between people must be treated with the utmost seriousness.
A key focus of the recruitment effort is reaching those on the “front lines” of potential exposure, such as poultry farmers, veterinarians, and agricultural workers. By shifting the trial from traditional hospital settings into community clinics, the NIHR aims to gather data from a diverse cross-section of the population most likely to encounter the virus. Beyond the scientific objectives, the study also includes a significant global equity component. Under an agreement with the Coalition for Epidemic Preparedness Innovations (CEPI), Moderna has committed to allocating 20% of its manufacturing capacity to supply low- and middle-income countries at affordable prices should a pandemic be declared.
As the seven-month trial progresses, the data will be used to inform future public health responses and build a “library” of vaccine blueprints for emerging threats. Professor Lucy Chappell, CEO of the NIHR, described the launch as a testament to the power of strategic partnerships and modern research infrastructure, noting that the trial setup time was halved compared to previous national targets. While the world hopes that H5N1 never achieves the mutations necessary to trigger a global crisis, the commencement of these trials ensures that for the first time in history, humanity may be ready with a solution before the problem fully arrives.