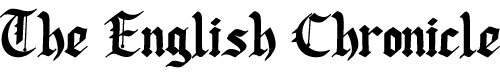Published: 21th July ‘2025 | The English Chronicle Online Desk
As global temperatures climb and public awareness of sun protection grows, a quiet revolution has been taking place in our bathroom cabinets and beach bags. Mineral sunscreens — often seen as the “natural” or “safer” option — are experiencing a boom in popularity, driven largely by concerns over health, environmental safety, and long-term skin damage. But beneath the glossy packaging and well-meaning marketing, a complex scientific debate simmers. Are mineral sunscreens really better than chemical ones, or are we simply misled by terminology and misunderstanding?
At the heart of the confusion lies the language we use. The distinction between “chemical” and “mineral” sunscreens often appears to be clear-cut, yet in reality, it is riddled with inaccuracies. Everything, after all, is made of chemicals — including water, zinc oxide, and even the air we breathe. According to Brian Diffey, emeritus professor of photobiology at the University of Newcastle and the mind behind the sunscreen UVA star rating system, what we commonly call “chemical” sunscreens are better described as “organic,” meaning they are carbon-based compounds. In contrast, “mineral” sunscreens — typically composed of titanium dioxide and zinc oxide — are “inorganic,” as they do not contain carbon-hydrogen bonds. Both are chemicals; the distinction lies not in their danger or purity but in their molecular structure.
Still, the idea that mineral sunscreens are safer continues to gain ground, spurred by a flurry of reports suggesting chemical filters like oxybenzone or avobenzone may disrupt hormones, damage coral reefs, or cause allergic reactions. As a result, shoppers are increasingly reaching for products labelled “reef-safe” or “natural.” Yet, most of the fears surrounding chemical sunscreens remain unproven or exaggerated. Regulatory agencies around the world, including the US Food and Drug Administration and the EU’s Scientific Committee on Consumer Safety, continue to approve these filters as safe for human use when applied correctly.
Historically, humans have always sought ways to shield themselves from the sun. Long before modern SPF numbers became common, people relied on physical barriers like wide-brimmed hats, umbrellas, and thick oils or pastes. Ancient civilizations — from the Himba of Namibia who use ochre-based mixtures, to the Romans who slathered olive oil — understood the importance of sun protection. It wasn’t until the 19th century that scientists discovered ultraviolet radiation and found that certain naturally occurring substances could absorb these harmful rays. By the 1930s, a host of plant-based compounds like aesculin and tannins were being used to protect the skin. These, too, were “chemical” sunscreens by today’s definitions — organic compounds that absorb UV radiation.
The arrival of modern laboratory-made UV filters took sun protection to a new level. Chemicals like avobenzone and octinoxate were not only more effective but also more photostable, meaning they offered better and longer-lasting protection against both UVA and UVB rays. At the same time, zinc oxide and titanium dioxide began to be incorporated into products marketed as “mineral” or “physical” sunscreens — based on the assumption that they worked by reflecting sunlight away from the skin.
That assumption, however, has been thoroughly debunked. While it is true that early 20th-century regulatory documents, including a now-outdated FDA monograph from the 1970s, described mineral sunscreens as reflecting UV rays like tiny mirrors, modern scientific studies have demonstrated otherwise. According to a 2015 peer-reviewed study that meticulously examined the behaviour of inorganic UV filters, titanium dioxide and zinc oxide actually absorb 95% of the UV radiation they encounter. Only around 4-5% is scattered — not reflected. The notion that mineral sunscreens are “physical blockers” that function like umbrellas is a myth, albeit a persistent one.
Antony Young, professor emeritus of experimental photobiology at King’s College London, reinforces this point. He has spent decades studying the efficacy of sunscreen ingredients and emphasizes that both mineral and chemical filters primarily absorb UV radiation. Even that small portion of scattered light does not bounce off the skin in mirror-like fashion. Instead, it enters the substance, interacts with particles, and emerges again in different directions — a phenomenon known as scattering, not reflection.
Adding to the complexity is the fact that many commercial sunscreens marketed as “mineral” are actually hybrids. They contain a combination of organic and inorganic filters, carefully engineered to achieve a balance between cosmetic appeal, water resistance, and broad-spectrum coverage. This blending of ingredients blurs the already fuzzy boundary between the two camps, making simple classifications increasingly obsolete.
Ultimately, the choice between mineral and chemical sunscreens may come down to personal preference, skin type, and cosmetic feel. Some people with sensitive skin may tolerate zinc oxide better, while others prefer the lightweight texture of avobenzone-based formulas. But as the science makes clear, no category has a monopoly on safety or effectiveness. What truly matters is that sunscreen — of any kind — is applied liberally, regularly, and correctly.
As consumers, it is vital that we move beyond misleading labels and marketing jargon to understand the actual science behind sun protection. Both mineral and chemical sunscreens have a role to play in protecting our skin from the harmful effects of ultraviolet radiation. Rather than vilifying one or championing the other on the basis of outdated or misunderstood claims, the best sunscreen is ultimately the one you’ll use — and reapply — every single day.
— The English Chronicle Online Desk