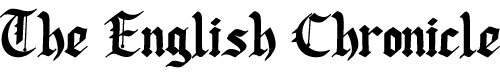Published: 02 May 2026. The English Chronicle Desk. The English Chronicle Online
A major breakthrough in global child health has been announced after the World Health Organization approved the first-ever malaria treatment specifically designed for newborn babies, marking what experts are calling a “major public health milestone” in the fight against one of the world’s deadliest diseases.
The treatment, known as Coartem Baby, has received prequalification from the World Health Organization (WHO), confirming that it meets international standards for quality, safety, and effectiveness. The approval is expected to allow rapid rollout across malaria-endemic regions, particularly in sub-Saharan Africa, where the disease remains a leading cause of infant and child mortality.
Malaria continues to pose a severe global health threat, with an estimated 610,000 deaths recorded in 2024 alone. According to health data, around three-quarters of those deaths occurred among children under the age of five, most of them in Africa. In some regions, up to 18% of infants under six months are believed to contract the disease.
Until now, babies suffering from malaria have typically been treated with medicines designed for older children, increasing the risk of incorrect dosing and potentially harmful side effects. Health experts have long warned that the lack of infant-specific treatment created a dangerous gap in global malaria care.
Coartem Baby has been developed to address that gap. The treatment can be safely used in infants weighing as little as 2kg (4.4lb) and comes in a sweet cherry-flavoured tablet form that can be dissolved in liquid, including breast milk, making it easier to administer to newborns.
The medicine contains a combination of two antimalarial compounds, artemether and lumefantrine. It was jointly developed by pharmaceutical company Novartis and the Medicines for Malaria Venture (MMV), a global non-profit partnership focused on malaria research and treatment development.
Dr Tedros Adhanom Ghebreyesus, director-general of the WHO, described the approval as a turning point in global health efforts. He said that for centuries malaria had devastated families and communities, but advances in treatment, vaccines, and prevention tools were now beginning to change that trajectory.
Medical experts involved in the development of Coartem Baby said the approval corrects a long-standing misconception that very young infants were naturally protected from malaria due to immunity passed from their mothers. Recent research has shown that newborns are, in fact, vulnerable to infection and require dedicated treatment.
Dr Martin Fitchet, chief executive of MMV, called the decision a “major public health milestone”, noting that infants had historically been excluded from effective malaria treatment options because existing medicines were not designed for them.
The urgency of the breakthrough is underscored by real-world cases in malaria-endemic countries. In Ghana, early access to the treatment has already begun. One infant, known as Baby Wonder, received Coartem Baby at just 12 weeks old after being hospitalised with a high fever and confirmed malaria infection. Doctors reported that the child recovered successfully following treatment.
Healthcare workers in affected regions say the availability of a safe infant formulation will significantly improve clinical outcomes. Dr Emmanuel Aidoo, a paediatrician at Methodist Hospital in Ankaase, Ghana, said the new drug provides much-needed confidence for doctors treating newborns with malaria for the first time.
“Having a treatment designed specifically for infants changes everything,” he said, highlighting how previously medical teams often had limited guidance when treating very young patients.
The pharmaceutical company Novartis has stated that it plans to make Coartem Baby available on a largely not-for-profit basis in regions where malaria is most prevalent, a move expected to increase access in low-income countries.
Global health organisations have welcomed the development as part of a broader shift in malaria control, alongside advances in vaccines, diagnostic tools, and next-generation mosquito nets. Together, these interventions are helping to reduce infection rates in some of the world’s most vulnerable populations.
Supporters of the programme also point to the importance of continued funding and international cooperation. The Medicines for Malaria Venture has received backing from multiple global donors, including the Gates Foundation, which has long supported malaria research and child health initiatives.
Health experts caution, however, that while the approval is a major step forward, malaria remains a persistent global challenge. Climate conditions, mosquito resistance, and gaps in healthcare infrastructure continue to complicate eradication efforts, particularly in rural and low-resource settings.
Nevertheless, the introduction of an infant-specific treatment represents a significant advancement in addressing one of the most overlooked areas of malaria care. By closing the treatment gap for newborns, health officials hope to reduce mortality rates further and strengthen long-term control of the disease.
As countries prepare to roll out Coartem Baby in national health systems, the WHO says the focus will now shift to ensuring equitable access, training healthcare workers, and integrating the new treatment into existing malaria programmes.
For millions of families in high-risk regions, the development offers renewed hope that even the youngest and most vulnerable children will finally have access to safe and effective malaria treatment.