Published: 10 September 2025 | The English Chronicle Desk
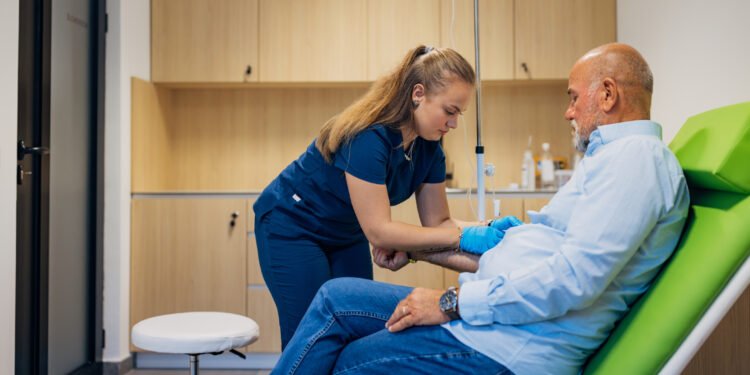
Doctors across the UK have launched a groundbreaking clinical trial for a £100 blood test designed to detect Alzheimer’s disease, aiming to transform how the NHS diagnoses and manages the condition. The trial seeks to recruit over 1,000 patients with suspected dementia from memory clinics nationwide to evaluate whether the test leads to faster, more reliable diagnoses and improved care for those affected.
Alzheimer’s disease, the most common form of dementia, affects more than half a million people in the UK, yet diagnosis remains a lengthy and complex process. Currently, only around 2% of patients undergo gold-standard tests such as PET scans or lumbar punctures that identify hallmark signs of the disease. Early and accurate detection is crucial, particularly as new drugs like lecanemab and donanemab, while only modestly effective, show greater promise when administered in the earliest stages of the condition.
The blood test being trialled measures a protein known as p-tau217, which reflects the presence of amyloid and tau—two proteins closely linked to Alzheimer’s. Early evidence indicates the test could match the accuracy of PET scans and lumbar punctures while being far less invasive and significantly more cost-effective.
Prof Jonathan Schott, neurologist at University College London and co-lead of the Adapt trial, said: “We’re expecting numerous further treatments to come on stream within the next 10 years, so we need to prepare the NHS now, to be able to deliver these treatments, as well as the benefits an accurate diagnosis already gives.”
Participants in the trial will come from diverse geographic, ethnic, and socioeconomic backgrounds, including those with other medical conditions, ensuring the findings are widely applicable. Half of the participants will receive their blood test results within three months, while the remainder will receive results after 12 months. Doctors will then assess whether access to these results improves diagnosis speed and guides treatment decisions.
Dr Ashvini Keshavan, senior clinical research fellow at UCL’s Dementia Research Centre and co-lead on the trial, explained: “This is not about confirming accuracy, which we have already done. This is about showing that it actually makes a difference to patients’ onward management.”
In laboratories already using the test, results can be processed within two weeks, highlighting the potential for rapid, large-scale implementation if the trial proves successful. Researchers expect to complete recruitment within two years, after which the results will be submitted to the National Institute for Health and Care Excellence (NICE) to determine whether the test should be rolled out across the NHS.
The trial is part of the multimillion-pound Blood Biomarker Challenge, funded by Alzheimer’s Society, Alzheimer’s Research UK, and the People’s Postcode Lottery. Dr Sheona Scales, director of research at Alzheimer’s Research UK, noted: “The earlier we can intervene with these experimental or new potential treatments, the greater their chances of success. The pace of change in Alzheimer’s research is accelerating, and accurate, early diagnosis is key to unlocking these advancements.”
If successful, the £100 blood test could not only speed up diagnosis but also guide treatment decisions more effectively, potentially transforming care for thousands of people living with Alzheimer’s in the UK.